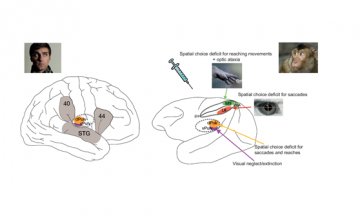
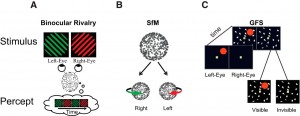
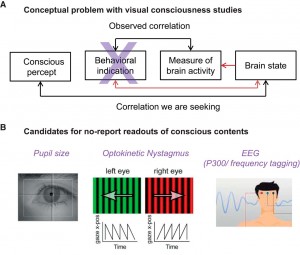
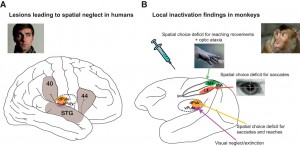
How does our subjective experience emerge from the brain? How does consciousness relate to the physical world? These age-old, deep questions are now at last being addressed directly and broadly by neuroscience and will become crucial in the decades to come.
The spectrum of consciousness-related conundrums is rapidly expanding: we are saving islands of human brain from devastating injuries, growing cerebral organoids in a vat, and building intelligent machines that perform faster and better than any healthy subject, just to mention a few.
Where does consciousness arise? Where is the boundary between insentient matter and a spark of subjectivity? Society needs to be scientifically and culturally prepared to face these emerging questions.
To do so, an approach with the broadest scope is needed: diverse theoretical frameworks, brain anatomy, physiology, and chemistry across scales and species, detailed and large-scale computer simulations, deep learning, neuromorphic computing, robotics, clinical neurology, anaesthesiology, psychology, behavioural, computational, and philosophical analysis must interact and blend on a single infrastructure.
These ingredients are naturally present in the Human Brain Project, and this workshop, stirred by world-leading scholars in the field, is where they come together for the first time.
Registration details will be available in the New Year.
Draft program and preliminary list of external speakers (further speakers and final program to be confirmed)
June 21, AM:
(1) Fundamental aspects, including theories of consciousness (Day1; 9-13):
– Larissa Albantakis: Dept of Psychiatry, University of Wisconsin-Madison, USA.
– Ned Block: Department of Philosophy, New York University, USA.
– David Chalmers: Department of Philosophy, New York University, USA + Centre for Consciousness, Australian National University, Australia.
June 21, PM:
(2) Neurobiological mechanisms and correlates of consciousness (Day1; 14 -18):
– Catherine Tallon-Baudry: Cognitive Neuroscience Laboratory, Institut National de la Santé et de la Recherche Médicale (INSERM)-École Normale Supérieure (ENS), Paris, France.
– Wolf Singer: Department of Neurophysiology, Max Planck Institute for Brain Research Frankfurt am Main, Germany ; Ernst Strüngmann Institute for Neuroscience in Cooperation with Max Planck Society Frankfurt am Main, Germany ; Frankfurt Institute for Advanced Studies, Johann Wolfgang Goethe University Frankfurt am Main, Germany.
– Nao Tsuchiya: School of Psychological Sciences, Monash University, Clayton, Australia + Monash Institute of Cognitive and Clinical Neuroscience, Monash University, Clayton, Australia.
– Rodolfo Llinas: Neuroscience Institute, Departments of Physiology, Neurology, and Psychiatry, and Center for Neural Science, New York University, New York, USA.
June 22, AM:
(3) Models, simulations, and emulation of consciousness (Day2; 9-13):
– Sean Hill: Neuroinformatics division, Campus Biotech, Geneva, Switzerland + Laboratory for the Neural Basis of Brain States, École Polytechnique Fédérale de Lausanne, Switzerland.
– Karl Friston: Wellcome Trust Centre for Neuroimaging, Institute of Neurology, University College London, London, UK.
– Fabrice Wendling: INSERM, U642, Université Rennes 1, Rennes, France.
June 22, PM
(4) Clinical, ethical, and societal implications of consciousness research (Day2; 14 -18):
– Nicholas Schiff: Feil Family Brain and Mind Research Institute + Dept of Neurology, Weill Cornell Medical College, New York, USA.
– Emery Brown: Department of Anesthesia, Critical Care and Pain Medicine, Massachusetts General Hospital, Harvard Medical School, Boston, MA, USA + Department of Brain and Cognitive Science, Massachusetts Institute of Technology, Cambridge, MA, USA.
– Melanie Wilke: Department of Cognitive Neurology, University Medicine Goettingen, Goettingen + German Primate Center, Leibniz Institute for Primate Research + German Research Foundation (DFG) Center for Nanoscale Microscopy and Molecular Physiology of the Brain (CNMPB), Georg-August-Universitaet Goettingen, Goettingen, Germany.
– Olaf Blanke: Laboratory of Cognitive Neuroscience, Brain-Mind Institute, Ecole Polytechnique Fédérale de Lausanne, Lausanne, Switzerland + Center for Neuroprosthetics, Ecole Polytechnique Fédérale de Lausanne, Lausanne, Switzerland.